CLIP-170S is a microtubule +TIP variant that confers resistance to taxanes by impairing drug-target engagement - ScienceDirect

Structural basis for tubulin recognition by cytoplasmic linker protein 170 and its autoinhibition | PNAS

CLIP-170S is a microtubule +TIP variant that confers resistance to taxanes by impairing drug-target engagement - ScienceDirect

Ninein is essential for apico-basal microtubule formation and CLIP-170 facilitates its redeployment to non-centrosomal microtubule organizing centres | Open Biology

Interactions between CLIP-170, Tubulin, and Microtubules: Implications for the Mechanism of CLIP-170 Plus-End Tracking Behavior | Molecular Biology of the Cell
The CLIP-170 N-terminal domain binds directly to both F-actin and microtubules in a mutually exclusive manner
CLIP-170S is a microtubule +TIP variant that confers resistance to taxanes by impairing drug-target engageme

α-Tubulin Tyrosination and CLIP-170 Phosphorylation Regulate the Initiation of Dynein-Driven Transport in Neurons - ScienceDirect

Quelle fonction pour la CLIP-170? : recherche de partenaires et nouveaux outils d'investigation | Semantic Scholar
CLIP-170S is a microtubule +TIP variant that confers resistance to taxanes by impairing drug-target engageme
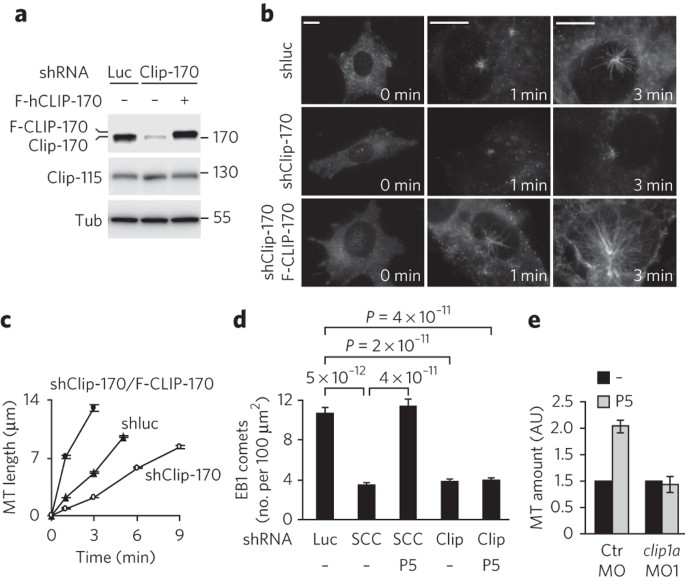
Pregnenolone activates CLIP-170 to promote microtubule growth and cell migration | Nature Chemical Biology

CLIP‐170 spatially modulates receptor tyrosine kinase recycling to coordinate cell migration - Zaoui - 2019 - Traffic - Wiley Online Library

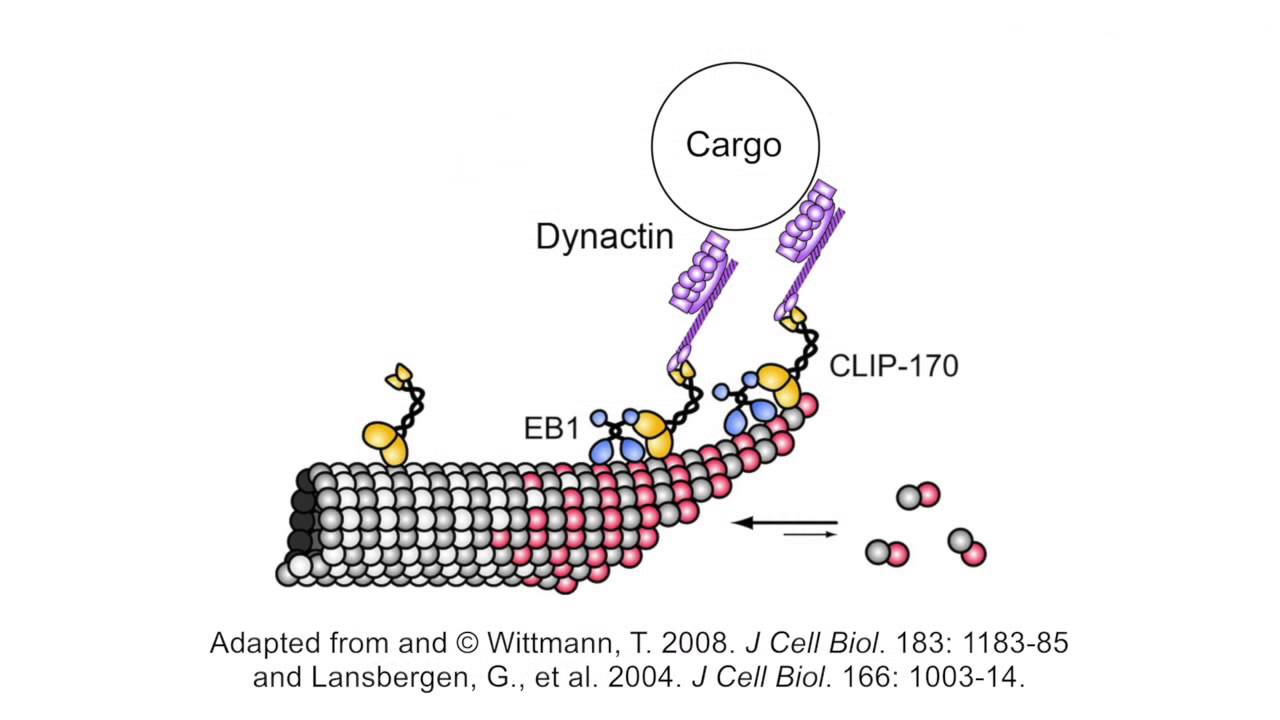
Microtubule binding proteins CLIP-170, EB1, and p150Glued form distinct plus-end complexes - ScienceDirect

Interactions between CLIP-170, Tubulin, and Microtubules: Implications for the Mechanism of CLIP-170 Plus-End Tracking Behavior | Molecular Biology of the Cell